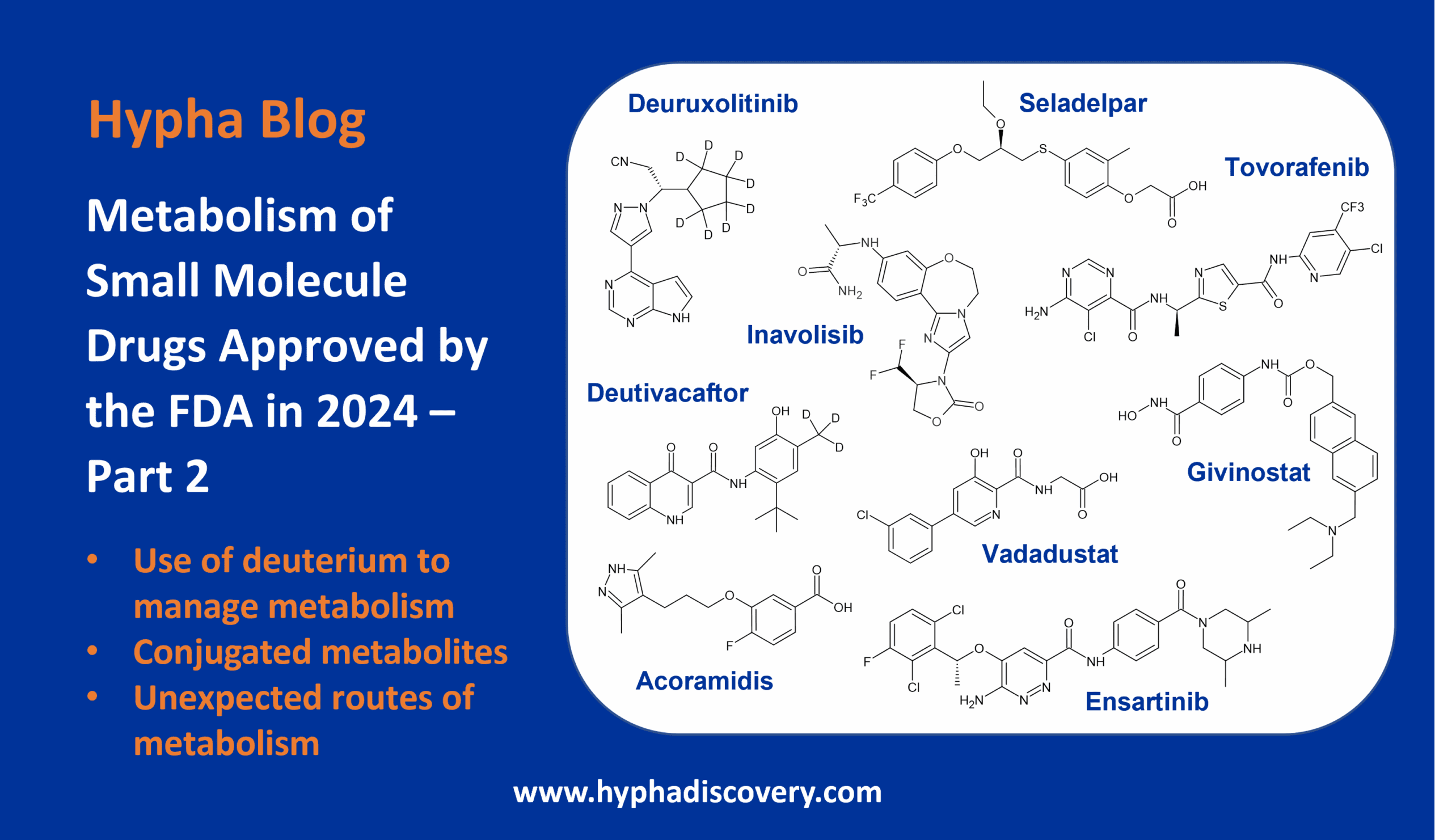
Metabolism and disposition of ozanimod in humans
Check out the implications of the complex metabolism of ozanimod discussed in a paper from Sekhar Surapaneni and other scientists at BMS. There are a plethora of elements involved in metabolism of the drug and implications concerning timing of metabolism studies:
- The uncovering of active major circulating and disproportionate human metabolites late in development
- The involvement of multiple non-CYP phase 1 mechanisms,
- The contribution of gut microbes
- Timing of metabolism studies
Critically, the authors use this story to highlight the importance of doing the radiolabelled human AME studies at the right time during drug development.
We highly recommend a read of this open access paper.

A commentary on the intricacies surrounding this are also discussed in a blog article written by Julia Shanu-Wilson for Drug Hunter – Billion-Dollar Biotransformations: On the Metabolism of Ozanimod.
Today, ozanimod is a likely megablockbuster drug approved for ulcerative colitis and multiple sclerosis. But before it became the first S1P modulator approved in ulcerative colitis, it received a scary, headline-making “Refuse to File” letter from the FDA due to insufficient preclinical characterization of an active metabolite RP112273 (CC112273). Addressing the refusal to file letter and FDA concerns around potential drug-drug interactions led to a two-year delay before ozanimod was approved and marketed, resulting in likely billions of dollars of lost sales and risking its leading position in the competitive S1P modulator space. The case study highlights the value of intricately understanding drug metabolism early in the drug discovery process. Read more.
Suspect involvement of gut metabolism?
If you suspect involvement of gut metabolism in disposition of your drug, Hypha can investigate this as part of our Gut Metabolites service. We have undertaken a number of studies for clients exploring metabolism of their drug and metabolites in a human faecal incubation system. A variety of biotransformations have been observed including reduction, hydroylsis, ring opening and conjugation.
References:
Ozanimod Human Metabolism and Disposition
Sekhar Surapaneni, Usha Yerramilli, April Bai, Deepak Dalvie, Jennifer Brooks, Xiaomin Wang, Julie V. Selkirk, Yingzhuo Grace Yan, Peijin Zhang, Richard Hargreaves, Gondi Kumar, Maria Palmisano and Jonathan Q. Tran
Drug Metabolism and Disposition May 1, 2021, 49 (5) 405-419; DOI: https://doi.org/10.1124/dmd.120.000220