Our Products
PolyCYPs+ Kits for Metabolites
PolyCYPs Metabolite Kits
Easy-to-use in-vitro enzyme system for generation of CYP, AO and FMO derived metabolites of drugs, agrochemicals and other small molecules
PolyCYPs+ metabolite kits are used by scientists in pharmaceutical and agrochemical companies to screen for and scale-up human and other mammalian phase I metabolites.
Hypha’s PolyCYPs enzymes are mined from talented biotransforming strains in Hypha’s microbial collection and expressed in E.coli together with ferredoxin and ferredoxin reductase redox partners. The enzymes have been rigorously tested for biocatalytic activity against a diverse panel of substrates and are capable of oxidising groups in a wide range of substrate compounds, including arylic/benzylic, -CH, -CH2, -CH3, –tert-butyl, and iso-propyl moieties, as well as aromatic systems for phenols and epoxides. De-alkylation of N– and O-alkyl moieties is also observed.
PolyCYP isoforms are available in kit form for clients to use in their own labs, or there is the option to outsource screening, scale-up and purification of PolyCYPs reactions to Hypha.
PolyCYPs+ Metabolite Screening Kits
PolyCYPs+ kits have a high success rate of producing human phase I metabolites. Each kit contains 18 PolyCYPs enzymes together with recombinant human aldehyde oxidase (AOX1) and the main human hepatic flavin-containing monooxygenase (FMO3). All the other human FMO isoforms are also available from Hypha for testing.
The kits have also been successfully used for making GSH conjugates by incubating the reactions in the presence of glutathione. Reactions can be scaled up to obtain material for structure elucidation by NMR to pinpoint the position of attachment.
Content of PolyCYP+ metabolite kits

All reagents are provided as lyophilised powders in sealed vials together with a 24-well reaction plate and air-permeable seal. Everything is included in the kit that you will need to perform the reaction in your lab, just add your compound and water!
18 PolyCYPs enzymes
AOX1
FM03
Cofactor recycling system containing glucose-6-phosphate (G6P), nicotinamide adenine dinucleotide phosphate (NADP+), and glucose-6-phosphate-dehydrogenase (G6PDH)
Formulation reagent to aid the solubilisation of test compounds of poor aqueous solubility
Positive control substrate (bosentan)
Extra enzyme (PolyCYP 6) for use with bosentan control reaction
PolyCYPs scale-up
PolyCYP enzyme reactions are fully scalable from mg to gram quantities through a number of routes, either by resupply of lyophilised enzymes for mg scale production in house, or larger scale production at Hypha, with optional purification and structure elucidation. We are also able to scale reactions derived from the human aldehyde oxidase (AOX1) and flavin-containing monooxygenase 3 (FMO3) contained in the PolyCYPs+ kit.
Multiple 10ml scale-up vials, suitable for converting 1mg of parent per vial, can be ordered off-the-shelf for any isoform for in-house scale-up in a kit format. Co-factors and formulant are also included along with a 24-well block suitable for performing 4 x 2.5 ml reactions. Other incubation formats can also be used, as recommended in the instructions.
N.B. Scale-up vials of lyophilised AOX1 are available, however we observe superior conversions using permeabilised whole cell reactions and thus it is recommended to scale-up AOX1 reactions at Hypha.
For larger quantities of any PolyCYPs+ products, we offer reactions undertaken at Hypha using cell paste from the recombinant E.coli or streptomycete clone containing the enzyme of interest. Contact us to find out more about this option.
Hypha also offer a service option should you prefer us to undertake scale-up reactions, or to process and purify metabolites from reaction extracts. We also have expertise in structure elucidation of products using cryoprobe NMR spectroscopy.
Case Studies - PolyCYPs in use
Use of PolyCYPs in radiolabelling studies
PolyCYPs metabolite kits can be used to generate radiolabelled CYP metabolites of drugs and agrochemicals for comparison with radio profiles from metabolism studies.
The benefits of this approach are two-fold: firstly, the cleaner matrix offered by PolyCYPs provides a less complex background against which to structurally characterise low-level metabolites by LC-MS/MS, which otherwise can be challenging especially for agrochemical matrices such as soil and plant extracts, animal faeces and bile.
Secondly, in situations where definitive identification by NMR is required, or purified metabolites are required for activity or liability testing, a PolyCYPs product matching the required metabolite can be scaled up to generate purified material for these applications, without prior knowledge of the metabolite structure.
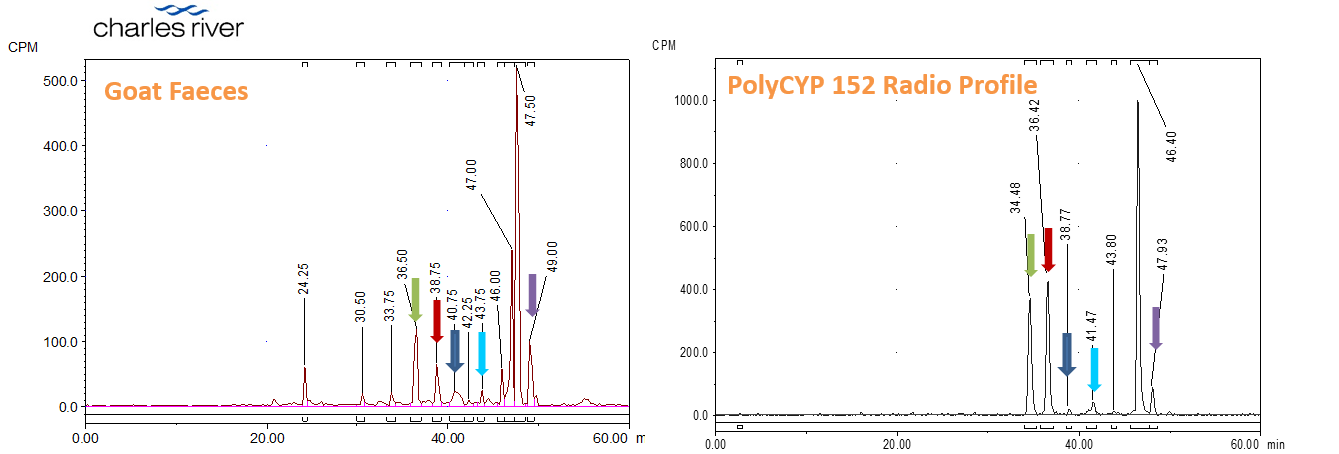
In this application a 14C-radiolabelled agrochemical was incubated with PolyCYPs enzymes and the metabolites produced compared with those detected in goat faeces as part of a mass balance study conducted by Charles River. High resolution LC-MS/MS showed oxidation of cycloalkyl and aromatic carbons of the compound by the PolyCYPs enzyme giving the best conversion for this compound. Metabolites in this PolyCYPs sample matched the radiolabelled metabolites in the goat faeces sample, thereby providing cleaner MS/MS data for interpretation in addition to an immediate route to access and scale-up the goat CYP-derived metabolites.
Synthesis of CYP3A4 metabolites using PolyCYPs
PolyCYPs kits can be used to screen for major phase I metabolites observed during preclinical and clinical studies, and scaled up to provide more material using scale-up vials.
In this case study, scientists at Boehringer Ingelheim used PolyCYPs kit to look for production of two hydroxylated metabolites of BI 894416. M398(2) is a major metabolite comprising 11.4% of total drug related exposure in the single ascending dose (SAD) study AUC0-24h pool. PolyCYP 152 best produced the major metabolite M398(2) at a conversion of 40% compared to the 5% observed in human liver microsomes (HLMs).
A 1mg scale-up using a PolyCYP 152 scale-up vial resulted in a 65% conversion to M398(2) to generate sufficient material for purification and structure elucidation by NMR. The position of hydroxylation was confirmed by NMR spectroscopy, including the stereochemistry, and was proven to be identical to the metabolite observed in HLM incubations. Further scale-up of the PolyCYP reaction was performed to generate milligram quantities for DDI and pharmacological activity studies.

Gram scale-up of a human disproportionate metabolite
Scientists at a US pharma company used a PolyCYPs+ metabolite screening kit in their own labs to identify a PolyCYP enzyme that gave a good conversion to the target human disproportionate metabolite. The client used scale-up vials to generate enough pure material for determining the structure by NMR.
Since the metabolite was not easy to chemically synthesise, further scale-up was outsourced to Hypha. Both the streptomycete clone and wild type strain from which the PolyCYP enzyme was cloned gave a high conversion to the target metabolite at a x5 increased dose of the parent, enabling multi gram amounts of the metabolite to be provided back to the client at >99% purity.

Why use PolyCYPs+ kits?
Quick access to human and other mammalian CYP and other phase I metabolites, including active metabolites
Applicable to a wide variety of drug, agrochemical and other small molecules
Easy-to-use – all reagents are provided as lyophilised powders, just add water
Discover new hydroxylated derivatives of a wide variety of drug compounds, generating potentially novel chemical matter in parallel
Comes with all reagents needed, including a 24-well reaction block and air permeable seal
Readily scalable - use in-house or outsource to Hypha
Resources
Explore our library of resources comprising brochures, case studies, posters and publications about the work we do.
In this case study at least 2 mg of a monohydroxylated metabolite (M4), originally observed in rat liver microsomes, was required by a US pharma company. Using PolyCYPs, the target metabolite was supplied to the client together with a Certificate of Analysis within 22 days from receipt of order, exemplifying the short timelines achievable using PolyCYPs to access CYP-derived metabolites.
Once a target metabolite or oxidised derivative has been synthesised by one or more PolyCYPs enzymes in the screening kit, a scale-up reaction with the best performing isoform is performed in order to access mg amounts of material for MetID and biological testing. The quickest and most cost-effective route for generating low mg amount of product is through the use of scale-up vials. Higher amounts of product can be generated using either a recombinant E.coli cell paste or through fermentation of a recombinant streptomyces clone expressing the isoform responsible for the biotransformation.
Hypha’s PolyCYPs kits are in routine use by pharma and agchem companies for producing human and other mammalian metabolites. One application involves use of PolyCYPs for creating radiolabelled metabolites for direct comparison with the radio profiles from mass balance and distribution study samples, necessary for regulatory filing. PolyCYPs provides a clean route for scalable access to more of the CYP-derived metabolites observed in these matrices, for definitive MetID and any tox studies deemed necessary. This is especially useful where low concentrations or unstable metabolites in the mass balance sample make structural identification difficult.
Related Services
We offer a number of solutions for synthesis of phase 1 and phase 2 metabolites using our one-stop metabolite shop approach
Chemical Synthesis
Late-stage chemical glucuronidation
Mammalian biotransformation
Panels of liver S9s / microsomes
Recombinant enzymes
PolyCYPs, hrCYPs, AOX, FMOs
Microbial biotransformation
Proven panels of bacteria and fungi
Gut metabolism
Human faecal incubations under anaerobic conditions
Purification & structure elucidation
COAs, acquisition / interpretation of NMR data

“Access of hydroxylated compounds through PolyCYPs has saved our company time and resources. We are sure that we will have more opportunities to work together in the future.”
Program Leader
European Pharma Company
Ready to begin? Our scientists are available to talk through your requirements
Hypha Discovery is a UK-based CRO supporting pharmaceutical and agrochemical companies worldwide through the production of metabolites and new derivatives of drugs and agrochemicals in discovery and development.
Resources
Cookie Policy | Privacy Policy | Website Terms and Conditions
© Hypha Discovery 2021. All Rights Reserved. Website by Fifteen.co.uk


