Metabolite Synthesis
Synthesis of Glucosides
Glucoside synthesis
Although glucosidation constitutes a relatively minor metabolic pathway in humans, the formation and urinary excretion of N-glucosides is important for some drugs such as barbiturates. In contrast, formation of glucoside metabolites of agrochemicals occurs more frequently in plants.
In plants, further metabolism of some glucosides leads to malonylated conjugates through the addition of a malonic acid residue to the sugar conjugate, so called secondary conjugation. A well known example is the formation of O-malonyl-N-glucoside of metribuzin. This conjugation pathway is critical for the tolerance of certain crops, such as tomatoes and soybeans, to metribuzin as conjugation converts the active herbicide into a harmless form.
Hypha have a number of techniques for glucoside synthesis. This includes microbial biotransformation and a number of glucosyltransferases cloned from microbes in our collection. In addition, our late-stage chemical synthesis techniques are effective at making these conjugates. Malonylated conjugates can be made using Hypha’s recombinant enzymes.
What We Do
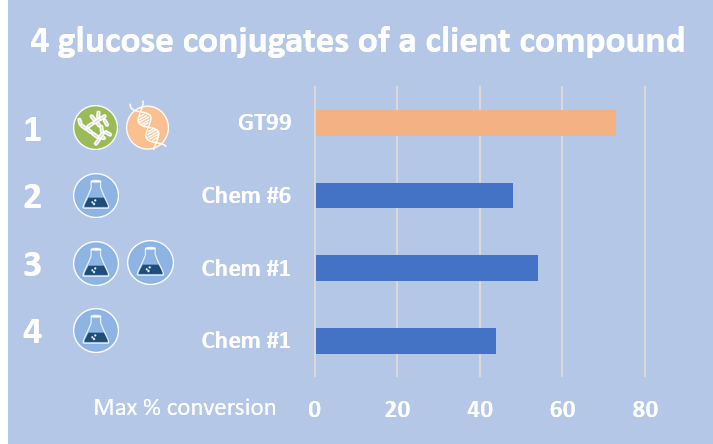
Case Study - Synthesis of N-glucosides of an agrochemical
In this project, four glucoside metabolites of an agrochemical were synthesised.
Three glucosides were synthesised by late-stage chemical synthesis, with the other made by both microbial biotransformation and recombinant glucosyl transferases. All of these were produced in good yield.
For the glucoside conjugate made using microbial biotransformation and glucosyltransferase enzymes, the best conversion was achieved using a mutant created from the parent glucosyltransferase, more than doubling the conversion to over 70%.
Chemical synthesis produced 3 different glucosides using two chemical conditions that produced the conjugates at good conversions.
Case Study - Synthesis of malonylated icariin
Malonyl glucosides can be observed as plant metabolites of herbicides (e.g.metribuzin, flurtamone), fungicides (e.g. fenhexamid, spiroxamine) and insecticides (e.g. carbofuran, fenpropathrin).
Hypha have cloned a number of proprietary glucosidating enzymes to first make the glucose conjugate. Accessing malonylated glucosides can present more of a challenge where a malonyl group is attached to a sugar moiety (usually glucose) in a compound. To achieve this, Hypha cloned and expressed the two relevant enzymes involved in E.coli and incubated them together with the substrate and co-factors. This is illustrated below using malonylation of icariin.

Find out about synthesis of other metabolite types
Resources
Explore our library of resources comprising brochures, case studies, posters and publications about the work we do.
Metabolites of some agrochemical products such as pesticides and herbicides possess greater plant or mammalian toxicity compared to the parent compound, thus necessitating a need for their identification, study and provision of analytical reference standards to meet regulatory requirements. Where a synthetic route is challenging, or the identity of a metabolite is unknown, creation of metabolites by microbial biotransformation is often a successful alternative due to similarities of xenobiotic metabolism in mammals, birds, fish, soil, and to some extent, plants.1 Furthermore, biocatalysis affords aromatic and aliphatic site-selectivity as well as regiocontrol of aromatic hydroxylation.2
Hypha’s PolyCYPs kits are in routine use by pharma and agchem companies for producing human and other mammalian metabolites. One application involves use of PolyCYPs for creating radiolabelled metabolites for direct comparison with the radio profiles from mass balance and distribution study samples, necessary for regulatory filing. PolyCYPs provides a clean route for scalable access to more of the CYP-derived metabolites observed in these matrices, for definitive MetID and any tox studies deemed necessary. This is especially useful where low concentrations or unstable metabolites in the mass balance sample make structural identification difficult.
We have extensive experiences in synthesising glucuronides using either scalable biotransformation or proprietary late-stage chemical methods.

Hypha Discovery did a fantastic job synthesizing N- and O- glucuronides of our clinical stage drug substance. The project updates were detailed, our questions were answered in a timely manner, and the overall timeline was maintained. Hypha was highly recommended to us and I would not hesitate to recommend them to a colleague.
Joshua Day Ph.D, Director of Chemistry
Crestone, Inc., Colorado, USA
Ready to begin? Our scientists are available to talk through your requirements
Hypha Discovery is a UK-based CRO supporting pharmaceutical and agrochemical companies worldwide through the production of metabolites and new derivatives of drugs and agrochemicals in discovery and development.
Resources
Cookie Policy | Privacy Policy | Supplier Code of Conduct Policy | Website Terms and Conditions
© Hypha Discovery 2021. All Rights Reserved.


