Paper Pick – Strategic Use of Benzylic Alcohols Reveals Cryptic Hydrogen-Bonding Interactions: Discovery of HBC-12551 as a Potent Non-Covalent Bruton’s Tyrosine Kinase Inhibitor
We’re kicking off 2026 with a paper pick that beautifully highlights the role that hydroxyls and water networks can play in dramatically improving binding affinities at some targets.
The paper by scientists at Hansoh Bio describes how reorganisation of water networks in binding pockets can lead to extraordinary boosts in binding affinity and consequent enhanced cellular potency. As part of a scaffold-hopping program around the non-covalent BTK inhibitor pirtobrutinib, a carefully positioned benzylic alcohol was introduced to master this reorganisation, significantly boosting potency by displacing a key water molecule to form a hydrogen bond with the backbone NH of Cys481.
Benzyl alcohols have multiple advantages – they can serve as both a hydrogen bond donor and acceptor, and their free rotation allows them to capture hydrogen bonding interactions with optimal hydrogen bond angles. Introduction of this secondary benzylic alcohol here also addressed a chemical stability liability found in the previous lead compound.
To offset high efflux via transporters and improve microsomal stability, the free alcohol was capped with a small alkyl group without interfering with key hydrogen bonding interactions. This resulted in compound 31 as the final optimised lead compound with improved ADME properties.
Interestingly introduction of the methyl group displaced a water molecule resulting in an advantageous reorganisation of the hydrogen bonding network in the solvent-exposed pocket. This led to creation of an array of water molecules that interacted with amide and carboxylate moieties of multiple amino acid residues.
The resulting compound 31 inhibited both wildtype and C481S mutant BTK and exhibited robust antiproliferative effects in B-cell lymphoma cell lines and antitumor activity in vivo.
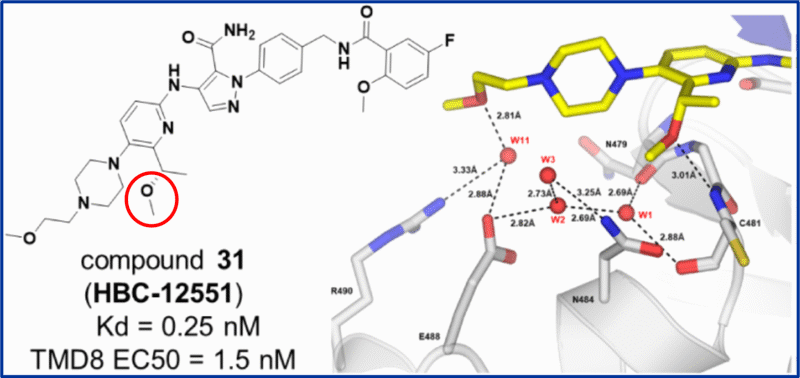
Water network illustration for compound 31 with key methoxy position circled (adapted from Liu et al., 2026)
In contrast to the rational design strategy employed by the authors, Hypha’s PolyCYPs enzymes can provide an empirical means to introduce hydroxyl groups in a late-stage way to compounds at the lead optimisation stage to help probe any potential gains in potency or other properties such as decreased lipophilicity, improved solubility and metabolic stability.
Paper
Liu et al. Strategic Use of Benzylic Alcohols Reveals Cryptic Hydrogen-Bonding Interactions: Discovery of HBC-12551 as a Potent Noncovalent Bruton’s Tyrosine Kinase Inhibitor. J. Med. Chem. 2026, 69, 3, 2867–2887. https://doi.org/10.1021/acs.jmedchem.5c0278I


