Metabolite Synthesis
Synthesis of CYP metabolites
CYP Metabolite Synthesis
CYP metabolite synthesis is possible using a variety of methods in our one-stop metabolite shop, including microbial biotransformation, mammalian biotransformation and using recombinant enzymes such as PolyCYPs and human recombinant CYPs.
Cytochrome P450 (CYPs) are monoxygenases that are involved in oxidative metabolism of a wide range of drugs and agrochemicals. They catalyse a variety of reactions including hydroxylation, epoxidation, N– and O-dealkylation and Bayer-Villiger oxidation. Various CYP isoforms are found in the liver and other tissues of the body, with the more prolific human CYP isoforms involved in drug metabolism comprising CYP3A4, CYP2D6, CYP2C9, CYP1A1/2, CYP2B6, CYP2C19, CYP2C8, CYP2A6, CYP2E1 and CYP2J2 (Zanger and Schwab, 2013).
CYP metabolite synthesis by whole cell microbial biotransformation
Wild-type microbes have broad oxidation capabilities due to the high number of CYP enzymes that can be expressed (25− 50 for bacterial species, 100+ for fungi). Hydroxylated and other oxidised derivatives can be produced, including aliphatic and aromatic hydroxylated derivatives.
What We Do
Case Study
Production of oxidised metabolites of ruxolitinib by microbial biotransformation
Incyte’s ruxolitinib is metabolised in humans largely by CYP3A4, and to a lesser extent by CYP2C9, to a number of oxidised metabolites. While it was possible for low milligram amounts of metabolites to be made using chemical synthesis, larger amounts were required for absolute stereochemical assignment by X-ray crystallography and for further biological testing.
Many microbes were able to produce some of the desired metabolites; however, one strain in particular was able to produce all the cyclopentyl 2-hydroxy- and 3-hydroxy metabolites plus the respective keto derivatives, and so was scaled up to enable the purification of up to 120 mg of each metabolite.
Several of these metabolites were found to possess pharmacological activity, including two metabolites resulting from hydroxylation at the 2- and 3-position on the cyclopentyl moiety, and the third a ketone resulting from further oxidation at the 3-position.
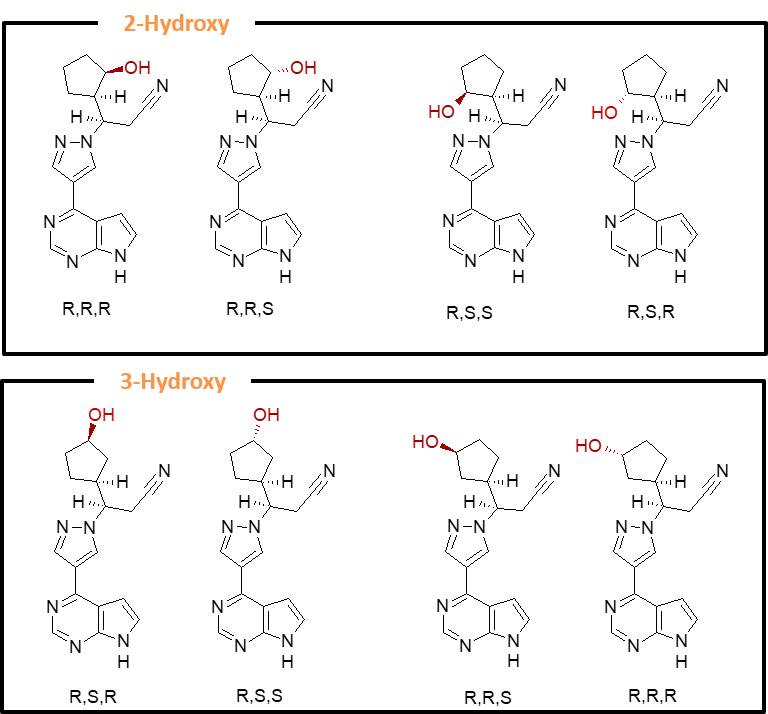
Hydroxylated CYP metabolites of ruxolitinib produced by Hypha’s microbial biotransformation system

CYP metabolites by PolyCYPs
Recombinant Cytochrome P450 enzymes can also be employed for CYP metabolite synthesis. These include PolyCYPs enzymes which have been mined from some of Hypha’s talented actinomycete bacteria and expressed in E.coli, providing a diverse set of CYPs effective for producing human and other mammalian CYP-mediated metabolites. They are soluble enzymes which are generally easy to express and scale-up. We also have a panel of human recombinant CYPs available.
PolyCYPs+ metabolite kits contain 20 enzymes effective for producing a wide range of phase 1 metabolites. In addition to 18 PolyCYPs enzymes, the kit also contains human aldehyde oxidase (AOX1) and the main human hepatic flavin-containing monooxygenase (FMO3), with the other human FMO isoforms also available at Hypha. Reactions are scalable either by resupply of lyophilized enzymes for mg scale production in-house, or larger scale production up to gram scale at Hypha, with optional purification and structure elucidation.
Resources
Explore our library of resources comprising brochures, case studies, posters and publications about the work we do.
Access a brochure on Hypha’s PolyCYPs+ kits. The kits contain 20 enzymes effective for producing a wide range of phase 1 metabolites. As well as 18 PolyCYPs enzymes, the kit also contains human aldehyde oxidase (AOX1) and the main human hepatic flavin-containing monooxygenase (FMO3), with other human FMO isoforms also available from Hypha.
Metabolite identification is an integral part of both preclinical and clinical drug discovery and development. Synthesis of drug metabolites is often required to support definitive identification, preclinical safety studies and clinical trials.
This poster illustrates the process undertaken at Hypha to produce a major human metabolite of ingenol disoxate for regulatory compliance.
Find out about synthesis of other metabolite types

Hypha Discovery has been a valuable metabolite ID partner. Hypha have provided biotransformation, metabolite purification and structure elucidation answers to some of our most challenging metabolism and metabolite ID problems. We really appreciate the breadth of expertise available at Hypha Discovery and will definitely reach out for future work.
Director of DMPK
US Pharma Company
Ready to begin? Our scientists are available to talk through your requirements
Hypha Discovery is a UK-based CRO supporting pharmaceutical and agrochemical companies worldwide through the production of metabolites and new derivatives of drugs and agrochemicals in discovery and development.
Resources
Cookie Policy | Privacy Policy | Website Terms and Conditions
© Hypha Discovery 2021. All Rights Reserved. Website by Fifteen.co.uk